VIABLE CLINICAL RESEARCH provides quality clinical trial infrastructure and services to CROs and Pharma. As the largest research management organization in Canada, we can fulfill all your clinical trial needs, including:
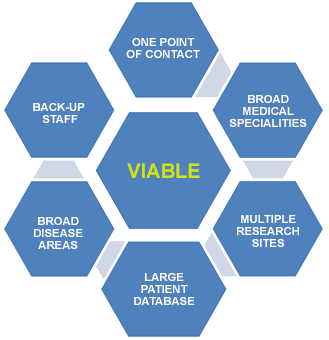
- One point of contact:
- For regulatory preparation and submission and
- For contract and budget negotiation
- Experienced CRCs on site, skills and certification in all study types
- Dedicated regulatory staff to handle all regulatory requirements
- Back-up staff for all sites to ensure adherence to timelines
- Standardized SOPs and site staff training
- Database of 100,000+ potential clinical participants
- Network of experienced investigators in numerous therapeutic areas
We work closely with your team to ensure successful trial execution!
In the clinical trial business since 2005, Viable is a reliable, reputable partner for your next clinical trial.
